Evidence-Based Timeline of Incretin-Driven Weight Loss
Introduction
In modern metabolic and aesthetic medicine, patients are increasingly interested not only in the effectiveness of therapy, but also in how quickly clinically visible results may appear. This question is particularly relevant for innovative treatments based on dual incretin receptor agonist therapies, which have well-documented metabolic and weight-management effects.
Omnitida BioActive, developed by BIOTECH LABORATORIUM LTD, is an advanced medication designed for medically supervised weight management. It integrates appetite regulation, metabolic optimisation, and long-term body composition improvement through dual incretin receptor activation.
Understanding the expected timeline of therapeutic response is essential for setting realistic expectations, improving adherence, and integrating pharmacological therapy into comprehensive aesthetic and medical treatment strategies.
Pharmacological Onset: Early Biological Activity
From a pharmacological perspective, dual incretin receptor agonist therapies begin exerting biological effects soon after administration. However, the onset of molecular activity should not be confused with the appearance of visible clinical results. While the medication interacts with incretin signaling pathways shortly after dosing, observable physiological changes develop gradually as metabolic processes adapt over time.
This progressive onset reflects the mechanism of dual receptor activation (GIP and GLP-1), which modulates appetite, insulin sensitivity, and energy balance rather than producing abrupt short-term weight changes.
Sources: pharmacodynamic analyses of dual incretin receptor agonist therapies; clinical pharmacology reviews on incretin-based treatments.
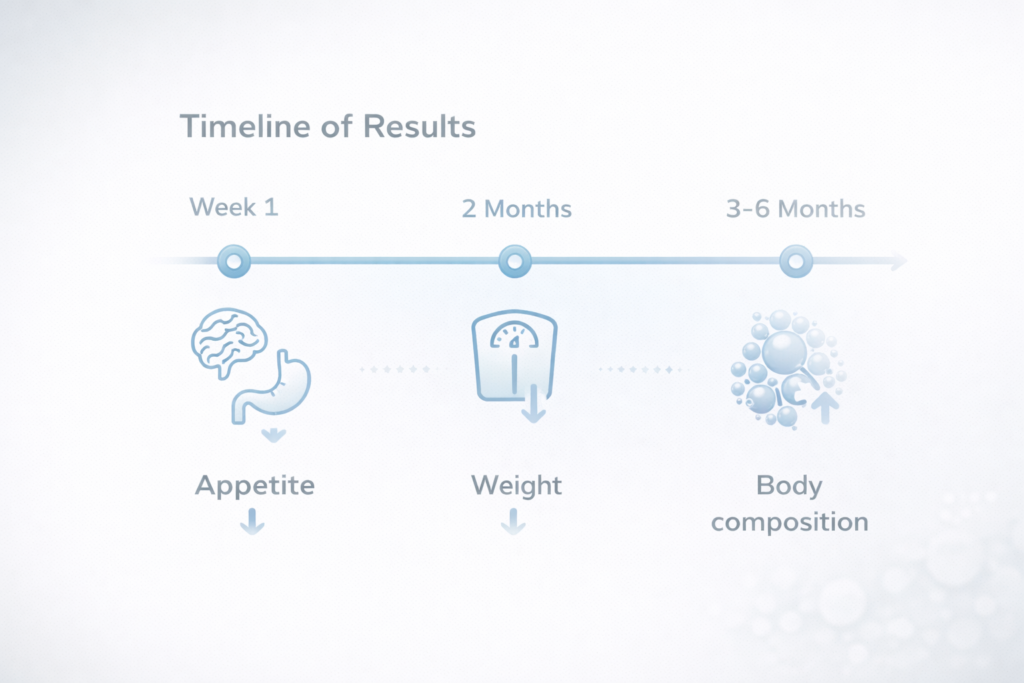
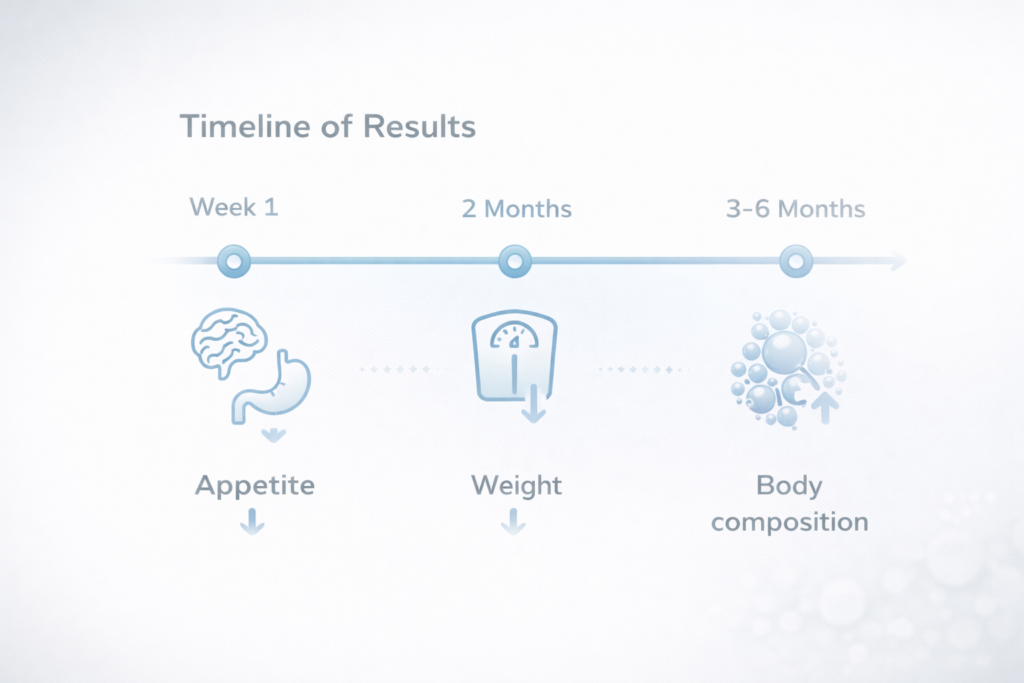
Weeks 1–2: Initial Appetite and Satiety Modulation

During the first one to two weeks of therapy, patients may begin to experience early neurohormonal effects associated with incretin pathway activation. These may include:
- Reduced hunger signals
- Earlier satiety during meals
- Decreased frequency of food cravings
These early responses are linked to central appetite regulation and represent the initial stage of behavioural and metabolic adaptation. Although significant weight change is not typically observed at this stage, this phase establishes the physiological basis for sustained caloric control.
Sources: early-phase clinical observations of tirzepatide; incretin-hormone appetite regulation studies.
Weeks 3–8: Initial Measurable Weight Reduction

Between approximately four and eight weeks of continued therapy, the metabolic effects of dual incretin–based treatment become increasingly measurable in clinical practice. Gradual weight reduction may begin to appear as a result of:
- Reduced caloric intake due to improved satiety
- Enhanced insulin sensitivity
- Improved metabolic efficiency
- Early reduction in visceral fat accumulation
Clinical trials of dual incretin receptor agonist therapies demonstrate that initial measurable decreases in body weight can emerge within the first two months of treatment, reflecting progressive rather than abrupt metabolic adjustment.
Sources: randomised controlled trials evaluating dual incretin receptor agonist therapies for obesity and metabolic disorders; interim clinical outcome analyses during the first 8–12 weeks.
Months 3–6: Clinically Significant and Visible Transformation
The most noticeable and clinically meaningful changes in body weight and composition are generally observed between three and six months of continuous therapy. During this period, patients may experience:
- Consistent and visible weight reduction
- Decrease in visceral adipose tissue
- Improvement in metabolic control
- Progressive reshaping of body composition
Large-scale clinical trials have demonstrated that dual incretin receptor agonist therapies produce substantial percentage reductions in baseline body weight over this timeframe, confirming the cumulative effect of sustained incretin modulation.
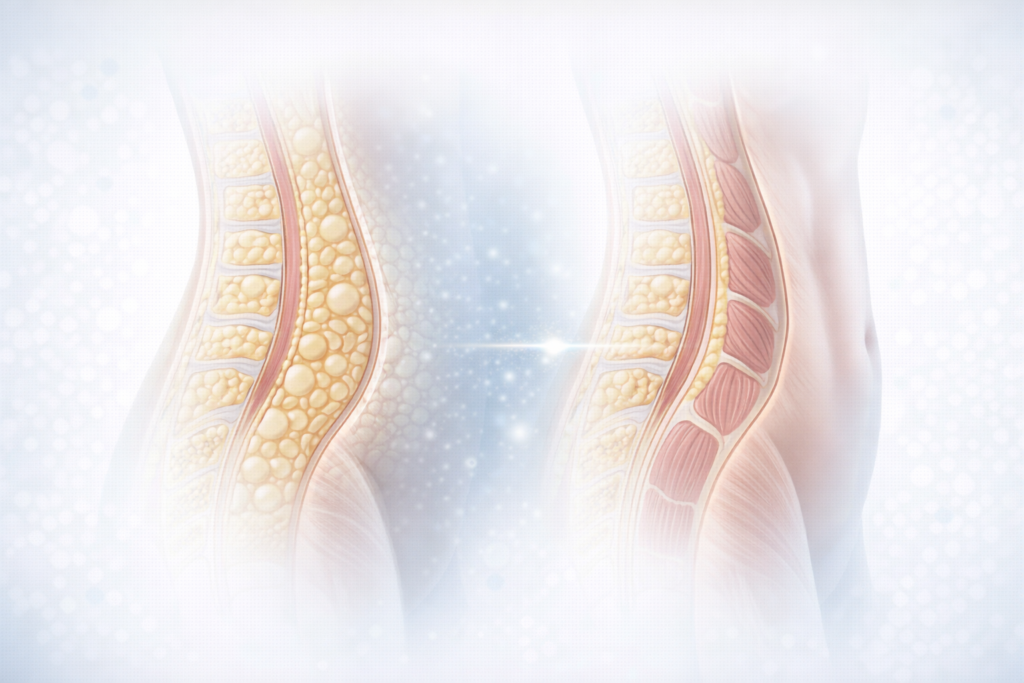
From an aesthetic-medicine perspective, this phase often coincides with improvements in body contouring outcomes, metabolic skin conditions, and overall appearance linked to systemic metabolic optimisation.
Sources: phase III clinical trials on dual incretin receptor agonist therapies; longitudinal analyses of weight-loss trajectories in metabolic medicine.
Long-Term Phase: 6–18 Months and Sustained Outcomes
Long-term evidence indicates that dual incretin receptor agonist therapies continue to promote progressive and sustained weight reduction when treatment is maintained under medical supervision. Over extended treatment durations, average reductions in body weight of approximately 15–22% have been reported in clinical research populations.
In addition to continued fat-mass reduction, long-term therapy has been associated with:
- Maintenance of achieved weight loss
- Improved insulin sensitivity and glycaemic balance
- Reduction in cardiometabolic risk factors
- Stable long-term appetite control
Such sustained outcomes support the role of dual incretin–based therapy as a long-term metabolic management strategy rather than a short-term intervention.
Sources: long-duration randomised clinical trials; longitudinal extension studies evaluating sustained efficacy of dual incretin receptor agonist therapies.
Individual Variability in Response
Although clinical data provide general timelines, the speed of visible results with dual incretin receptor agonist therapycan vary between individuals. Factors influencing response include:
- Baseline body weight and metabolic status
- Dose titration schedule
- Dietary behaviour and physical activity
- Degree of insulin resistance
- Adherence to treatment protocols
Importantly, dual incretin–based therapy is characterised by steady and progressive weight reduction, which is considered clinically advantageous due to improved metabolic adaptation and long-term sustainability.
Sources: personalised medicine analyses; subgroup evaluations from multicentre tirzepatide trials.
Clinical Relevance in Aesthetic and Metabolic Practice
In integrative aesthetic medicine, excessively rapid weight loss may negatively impact skin elasticity and overall aesthetic harmony. The gradual and controlled timeline observed with dual incretin receptor agonist therapies aligns well with cosmetological principles that prioritise balanced fat reduction and metabolic stabilisation.
Through its dual incretin mechanism, this therapeutic approach supports:
- Controlled appetite regulation
- Progressive reduction of fat mass
- Improvement in insulin resistance
- Optimisation of body-contouring outcomes
These properties make dual incretin–based therapy, including Omnitida BioActive, highly relevant for comprehensive programmes that combine pharmacological weight management with dermatological and aesthetic interventions
Sources: interdisciplinary publications in endocrinology and aesthetic medicine; reviews on metabolic health and cosmetic outcomes.
Omnitida BioActive: Translating Clinical Evidence into Practice
Omnitida BioActive, developed by BIOTECH LABORATORIUM LTD, reflects the clinically established timeline of incretin-driven metabolic therapy. Its therapeutic concept is based on progressive metabolic regulation through dual incretin receptor activation rather than immediate weight suppression.
An evidence-aligned response timeline with Omnitida BioActive may include:
- Early appetite and craving control within the first weeks
- Initial measurable weight reduction within one to two months
- Visible and clinically meaningful body-composition changes after three to six months
- Sustained long-term weight reduction with continued therapy
This staged progression supports both metabolic health and aesthetic optimisation, aligning pharmacological efficacy with realistic clinical expectations.
Conclusion
Dual incretin–based therapy represents a scientifically validated approach to weight management characterised by gradual, progressive, and sustained clinical effects. Early appetite modulation may occur within weeks, measurable weight reduction follows in the first months, and the most significant improvements in body composition typically develop between three and six months of continuous therapy.
Omnitida BioActive, developed by BIOTECH LABORATORIUM LTD, embodies this evidence-based therapeutic paradigm. By targeting fundamental metabolic mechanisms rather than isolated symptoms, it offers a predictable, clinically grounded, and aesthetically compatible pathway toward effective and sustainable weight management under professional medical supervision.
Sources: peer-reviewed clinical trials evaluating dual incretin receptor agonist therapies; pharmacological reviews on dual incretin receptor agonists; longitudinal studies on metabolic and weight-management outcomes.