Insights from Type 2 Diabetes Research and the SURPASS Clinical Programme
Introduction

The development of incretin-based therapies has transformed the clinical management of obesity and metabolic disorders. Dual incretin receptor agonist therapies, targeting both glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptors, represent a significant innovation in this field due to their broad metabolic effects demonstrated across multiple clinical trials in patients with type 2 diabetes.
Omnitida BioActive, developed by BIOTECH LABORATORIUM LTD, is an innovative medication designed in accordance with these advances in metabolic medicine. Its therapeutic concept aligns with contemporary clinical research demonstrating that dual incretin modulation can significantly improve glycaemic control, body weight, and cardiometabolic risk factors.
This article provides a professional reinterpretation of evidence derived from clinical trial programmes investigating dual incretin receptor agonist therapies, with particular focus on their role in metabolic regulation and medically supervised weight management.
Sources: ClinicalTrialsArena review of dual incretin receptor agonist development; SURPASS clinical trial programme analyses; peer-reviewed clinical trial literature on incretin-based metabolic therapies.
Development Background: Dual Incretin Therapies in Type 2 Diabetes Research

Dual incretin receptor agonist therapies were initially developed as therapeutic options for individuals with type 2 diabetes, aiming to improve glycaemic control through incretin hormone modulation. Their dual receptor activity allows simultaneous stimulation of both GIP and GLP-1 receptors, which play complementary roles in glucose metabolism and appetite regulation.
Large-scale clinical development programmes, including global clinical trial initiatives involving thousands of participants with diverse metabolic profiles, have enabled robust evaluation of efficacy and safety across multiple treatment settings. These studies demonstrated that dual incretin receptor modulation can address hyperglycaemia, insulin resistance, and excess body weight simultaneously.
Sources: ClinicalTrialsArena overview of dual incretin receptor agonist development; global clinical trial programme documentation evaluating incretin-based metabolic therapies; endocrinology reviews on dual incretin receptor agonists.
Effects on Glycaemic Control
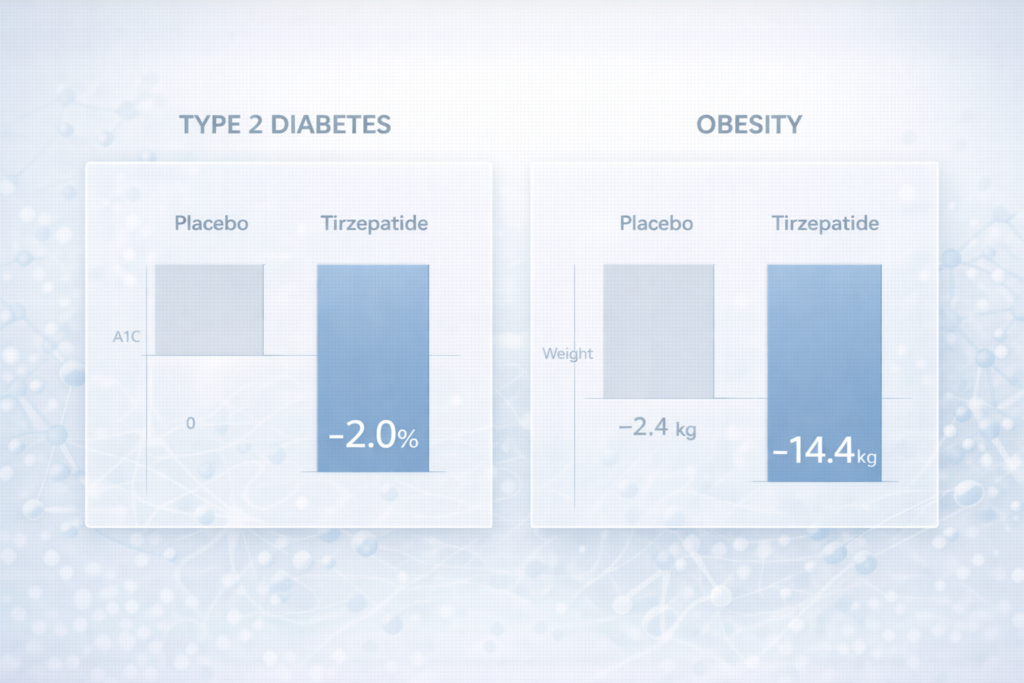
Clinical studies consistently demonstrate that dual incretin receptor agonist therapies produce substantial reductions in glycated haemoglobin (HbA1c), reflecting improved long-term glucose regulation. Phase 3 trials have reported reductions in HbA1c approaching approximately 1.9% to 2.6%, depending on dosage and patient population, confirming strong glucose-lowering efficacy.
A significant proportion of participants achieved recommended glycaemic targets during these trials, illustrating the therapy’s capacity to restore metabolic balance in individuals with type 2 diabetes. These effects are attributed to enhanced glucose-dependent insulin secretion combined with suppression of glucagon levels, which together improve overall metabolic homeostasis.
Sources: phase 3 clinical trials evaluating dual incretin receptor agonist therapies; pooled analyses of glycaemic outcomes with dual incretin modulation; endocrine research on incretin-mediated glucose regulation.
Impact on Body Weight and Body Composition
Beyond glycaemic control, clinical trials have shown that dual incretin receptor agonist therapies can lead to significant reductions in body weight, a critical therapeutic objective in both metabolic medicine and aesthetic practice. Across multiple studies, clinically meaningful reductions in body weight were observed alongside favourable changes in body composition.
These reductions were primarily associated with decreases in fat mass and improvements in energy balance mediated through appetite regulation and metabolic efficiency. The ability to influence both caloric intake and adipose tissue metabolism positions dual incretin–based therapy as a comprehensive weight-management strategy rather than solely a glucose-lowering intervention.
Sources: clinical trial analyses of weight-loss outcomes with dual incretin receptor agonist therapies; metabolic pharmacology reviews on incretin-based metabolic treatments; studies on adipose tissue response to incretin therapies.
Cardiometabolic and Systemic Benefits
In addition to glycaemic and weight outcomes, clinical trial evidence indicates that dual incretin receptor agonist therapies contribute to broader cardiometabolic improvements. Observed benefits include favourable effects on lipid profiles, blood pressure, and markers of systemic metabolic inflammation, all of which are closely associated with obesity-related morbidity.
These pleiotropic metabolic effects result from dual incretin receptor activation, which modulates multiple physiological pathways involved in energy homeostasis, insulin sensitivity, and adipose tissue function. Such systemic optimisation is particularly relevant for patients presenting with metabolic syndrome or insulin resistance.
Sources: multicentre analyses of cardiometabolic outcomes in clinical trials evaluating dual incretin receptor agonist therapies; reviews on dual GIP and GLP-1 receptor physiology; clinical endocrinology research on metabolic inflammation.
Quality of Life and Patient-Reported Outcomes
Clinical research also highlights improvements in patient-reported quality of life associated with dual incretin receptor agonist therapy. Participants in several trials reported better general health perception and improved weight-related quality-of-life scores following treatment, indicating that metabolic and anthropometric improvements translate into tangible benefits in everyday functioning.
These findings emphasise the importance of comprehensive metabolic improvement, not only for clinical parameters but also for long-term patient adherence and satisfaction with therapy.
Sources: patient-reported outcome analyses from multicentre clinical trials evaluating dual incretin receptor agonist therapies; quality-of-life assessments in metabolic medicine research.
Relevance for Integrative Aesthetic and Metabolic Medicine
From the perspective of aesthetic and dermatological practice, the metabolic improvements demonstrated in clinical trials are highly significant. Excess adiposity, insulin resistance, and systemic inflammation can adversely affect skin quality, body contour, and ageing processes.
By addressing these underlying metabolic drivers, dual incretin receptor agonist therapy supports:
- Gradual and controlled fat mass reduction
- Improved insulin sensitivity and hormonal balance
- Reduction of systemic metabolic inflammation
- Enhanced outcomes of body-contouring and dermatological interventions
These integrative benefits highlight the convergence of endocrinology and aesthetic medicine in modern personalised treatment strategies.
Sources: interdisciplinary research on metabolic health and dermatological outcomes; clinical evidence on adiposity, inflammation, and skin physiology.
Safety and Tolerability in Clinical Trials
Clinical trial data indicate that dual incretin receptor agonist therapies are generally well tolerated when administered under appropriate medical supervision. The most frequently reported adverse effects are gastrointestinal in nature and tend to occur during early dose-escalation phases, typically diminishing with continued treatment.
Careful dose titration and personalised monitoring remain essential to ensure optimal safety and therapeutic outcomes in routine clinical practice.
Sources: safety analyses from phase 3 clinical trials evaluating dual incretin receptor agonist therapies; systematic pharmacovigilance reviews on incretin-based therapies.
Omnitida BioActive: Translating Trial Evidence into Clinical Practice
Omnitida BioActive, developed by BIOTECH LABORATORIUM LTD, reflects the scientific and clinical findings observed across extensive clinical trial programmes evaluating dual incretin receptor agonists.
Its therapeutic model aligns with modern clinical understanding that effective weight management requires integrated regulation of appetite, insulin sensitivity, and adipose tissue metabolism rather than isolated glycaemic control alone.
Clinically relevant potential benefits include:
- Significant and sustained reduction in body weight
- Improvement in glycaemic balance and insulin resistance
- Reduction in visceral adipose tissue
- Positive influence on cardiometabolic risk factors
- Synergy with lifestyle modification and aesthetic treatment programmes
Conclusion
Evidence from large-scale clinical trials in patients with type 2 diabetes demonstrates that dual incretin receptor agonist therapies provide comprehensive metabolic benefits, including substantial improvements in glycaemic control, body weight, and cardiometabolic risk profiles. These findings underline the importance of dual incretin receptor activationas a modern therapeutic strategy in metabolic medicine.
Omnitida BioActive, developed by BIOTECH LABORATORIUM LTD, translates these clinically validated mechanisms into an innovative pharmacological solution suitable for integrative medical and aesthetic practice. Under professional supervision, dual incretin–based therapy offers a scientifically grounded, sustainable, and multidimensional approach to weight management and metabolic optimisation.
Sources: ClinicalTrialsArena overview of dual incretin receptor agonist development; phase 3 clinical trial programmes evaluating incretin-based metabolic therapies; peer-reviewed endocrinology and metabolic medicine literature on dual incretin receptor agonists.
