Evidence on efficacy, safety, and who was studied (Type 2 diabetes programme)
Introduction
When we talk about evidence-based metabolic therapy, the most practical question is often: what did the pivotal trials actually show, and who were the results based on? The US FDA “Drug Trials Snapshot” format is useful here because it summarises the core clinical-trial populations and headline outcomes that supported original approval.
Omnitida BioActive, developed by BIOTECH LABORATORIUM LTD, is an innovative medication based on dual incretin receptor activation. Below is a pharmacist- and aesthetic-medicine–oriented interpretation of the FDA trial snapshot data for dual incretin receptor agonist therapies, focusing on trial scale, efficacy signal, tolerability profile, and patient diversity—without discussing other branded medicines.
Sources: US FDA Drug Trials Snapshot for dual incretin receptor agonist injection therapies (original approval May 13, 2022; snapshot published June 13, 2023); FDA prescribing information referenced within the snapshot.

1) What Was Studied in the Pivotal Clinical Programme
In the FDA snapshot, dual incretin receptor agonist therapy is presented as a treatment intended to improve blood sugar control in adults with type 2 diabetes, used alongside diet and exercise. The primary efficacy measurement across the key trials was change in HbA1c from baseline (a standard marker of average glycaemic control over approximately three months).
From a metabolic and aesthetic standpoint, this is important because improved glycaemic control and reduced insulin resistance can influence visceral adiposity, inflammatory load, and skin-related metabolic effects that often accompany excess weight.
Sources: US FDA Drug Trials Snapshot (indication and endpoints for dual incretin receptor agonist therapy).
2) How large and globally representative were the trials?
The FDA snapshot reports that approval was supported by nine clinical trials including 7,769 adults with type 2 diabetes, with 5,415 participants receiving dual incretin receptor agonist therapy. Trials were conducted at 673 sites across 24 countries (including the United States and Puerto Rico, as well as multiple regions across Europe, Asia, and the Americas).
Safety pool: all nine trials (7,769 participants)
Efficacy pool: five of the trials, totalling 6,263 adult participants
This scope is meaningful for real-world confidence, as it indicates that findings were derived from a large multinational population and an extensive safety dataset, strengthening the generalisability of the clinical evidence.
Sources: US FDA Drug Trials Snapshot (trial counts, patient totals, site/country totals, efficacy vs safety populations).ug Trials Snapshot for tirzepatide (trial counts, patient totals, site/country totals, efficacy vs safety pools).
3) What the FDA summary says about effectiveness
Across the five pivotal efficacy trials, the FDA snapshot indicates that dual incretin receptor agonist therapy lowered HbA1c versus control. The snapshot tables show dose-dependent improvements, with comparator-adjusted HbA1c reductions typically in the range of about 1.2 to 1.7 percentage points, depending on dose and trial design (including whether background therapy was used).
The snapshot also states that effectiveness was similar across key demographic subgroups, specifically:
- Sex: similar in females and males
- Race: similar across the racial groups analysed
- Age: similar in those under 65 and those aged 65 and older
For clinical practice, that consistency across groups is important when counselling patients on expected metabolic response.
Sources: US FDA Drug Trials Snapshot (efficacy statement and subgroup consistency; HbA1c effect tables for dual incretin receptor agonist therapy).rug Trials Snapshot for tirzepatide (efficacy statement and subgroup consistency; HbA1c effect tables).
4) Safety and tolerability: what to expect most commonly
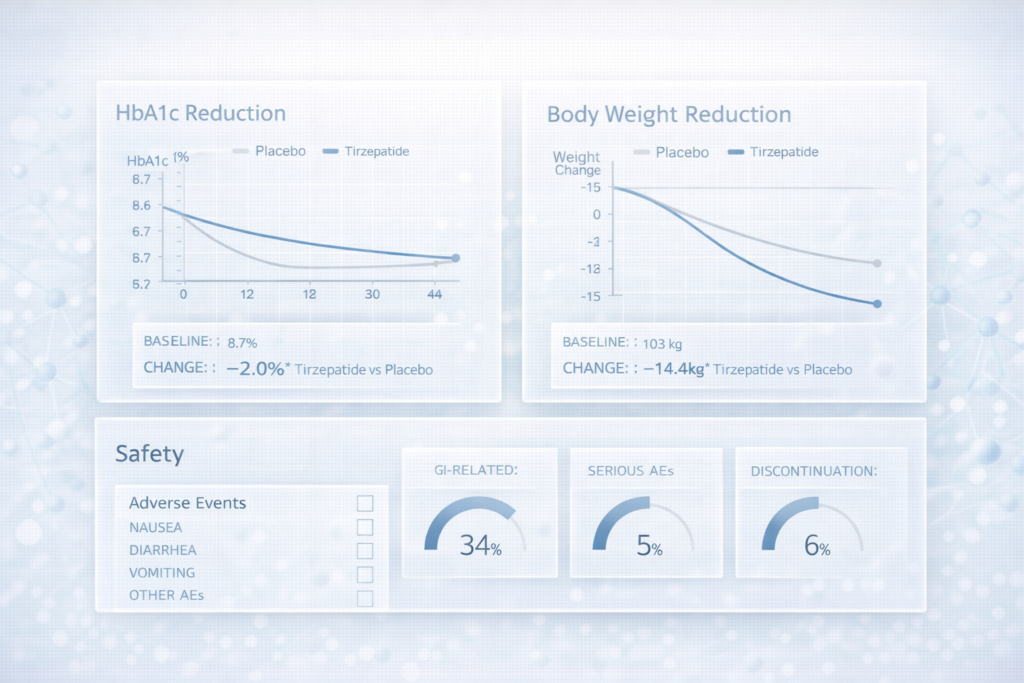
The FDA snapshot highlights gastrointestinal tolerability as the most frequent issue and lists the most common side effects observed in clinical trials as:
- nausea
- diarrhoea
- decreased appetite
- vomiting
- constipation
- indigestion (dyspepsia)
- abdominal pain
It also provides incidence rates (in two placebo-controlled datasets) showing a dose-related rise in several GI effects. For example (placebo vs higher doses, as reported in the snapshot):
- Nausea: 4% vs up to 18%
- Diarrhoea: 9% vs up to 17%
- Decreased appetite: 1% vs up to 11%
- Vomiting: 2% vs up to 9%
- Constipation: 1% vs up to 7%
Why this matters cosmetologically: rapid appetite reduction plus GI symptoms can lead to suboptimal protein intake and hydration in some patients—factors that influence skin turgor, hair cycle stability, and recovery from aesthetic procedures. In practice, this is where structured nutrition and gradual dose titration are not “nice to have”, but part of outcomes management.
Sources: US FDA Drug Trials Snapshot for dual incretin receptor agonist therapy (common adverse reactions table and common side effects list); FDA prescribing information referenced in the snapshot.
5) Serious safety warnings noted in the FDA snapshot

The snapshot lists serious risks that clinicians monitor for, including:
- pancreatitis
- hypoglycaemia (especially relevant when combined with other glucose-lowering therapies)
- allergic reactions
- kidney problems (including kidney failure)
- severe gastrointestinal problems
- complications of diabetic eye disease (diabetic retinopathy)
It also notes a thyroid tumour signal observed in rat studies for dual incretin receptor agonist therapies and related mechanisms, and that it is not known whether this translates to humans in the same way, which reflects the typical regulatory wording used for this therapeutic class.
For patient education, the key message is not fear but appropriate screening, counselling, and monitoring, particularly in individuals with risk factors or those receiving concurrent metabolic therapies.
Sources: US FDA Drug Trials Snapshot (serious side effects section for dual incretin receptor agonist therapy).
6) Who exactly was represented: demographics at a glance
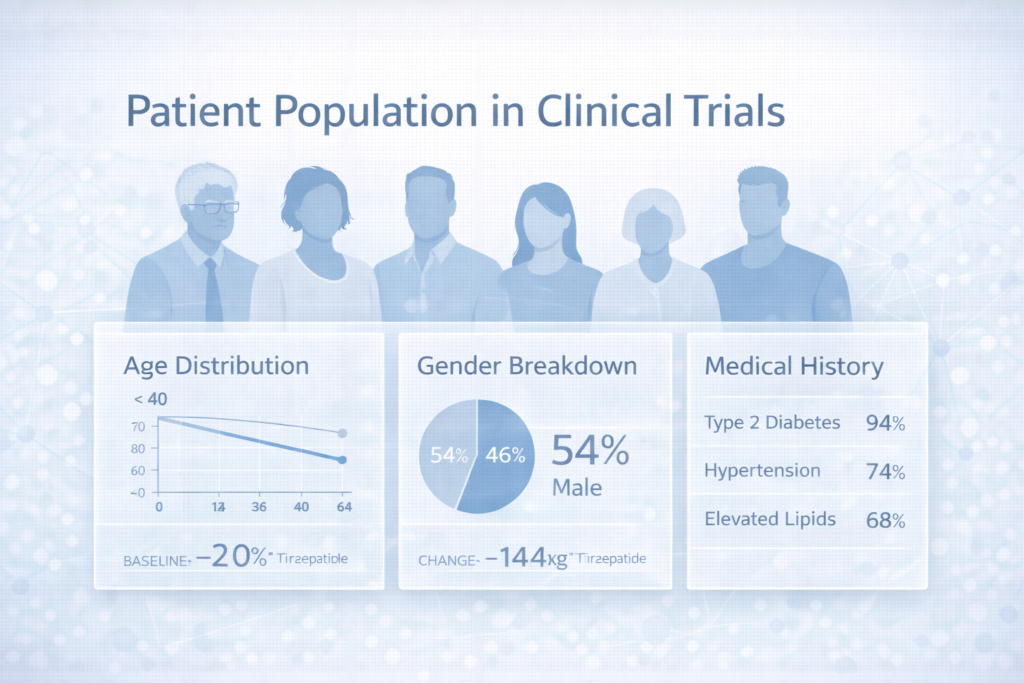
The FDA snapshot provides clear baseline demographics for the combined trial populations.
Efficacy population (five combined trials):
- Sex: 55% male (3,441), 45% female (2,822)
- Race: 80% White (5,037), 7% Asian (424), 4% Black/African American (224), 8% American Indian/Alaska Native (505), 1% Other (69)
- Age: 67% <65 years (4,181), 33% ≥65 years (~2,082)
Safety population (nine combined trials):
- Sex: 58% male (4,492), 42% female (3,277)
- Race: 69% White (5,337), 20% Asian (1,512), 3% Black/African American (269), 7% American Indian/Alaska Native (523), 1% Other (82)
- Age: 69% <65 years (5,331), 31% ≥65 years (2,438)
This is useful when you need to communicate what “evidence-based” means in concrete terms: large numbers, broad geography, and documented subgroup analyses.
Sources: US FDA Drug Trials Snapshot for tirzepatide (demographics snapshot figures and text).
How this translates to Omnitida BioActive in clinical positioning
Because Omnitida BioActive is based on dual incretin receptor activation, the FDA snapshot helps frame evidence-aligned expectations for:
- robust glycaemic improvement (HbA1c as a primary metabolic endpoint),
- predictable GI tolerability profile with dose-related frequency,
- consistent response across sex/age/race subgroups in the analysed datasets,
- clear monitoring priorities (pancreatitis symptoms, dehydration risk, renal function in susceptible patients, hypoglycaemia risk with certain combinations, and eye-disease monitoring in diabetes).
In integrated aesthetic practice, the most “practical” takeaway is that successful outcomes are often maximised by pairing metabolic therapy with:
- hydration and protein adequacy,
- skin-supportive micronutrition where appropriate,
- realistic pace-of-change counselling,
- and procedure timing that respects the patient’s adaptation phase.
