Clinical and Metabolic Insights from Contemporary Evidence
Introduction
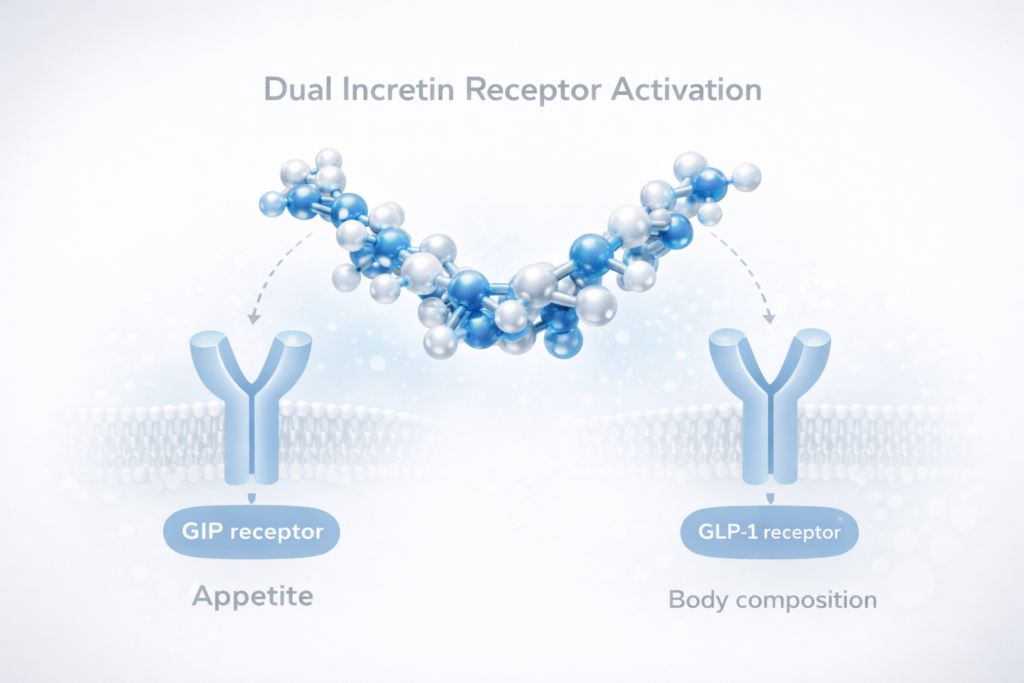
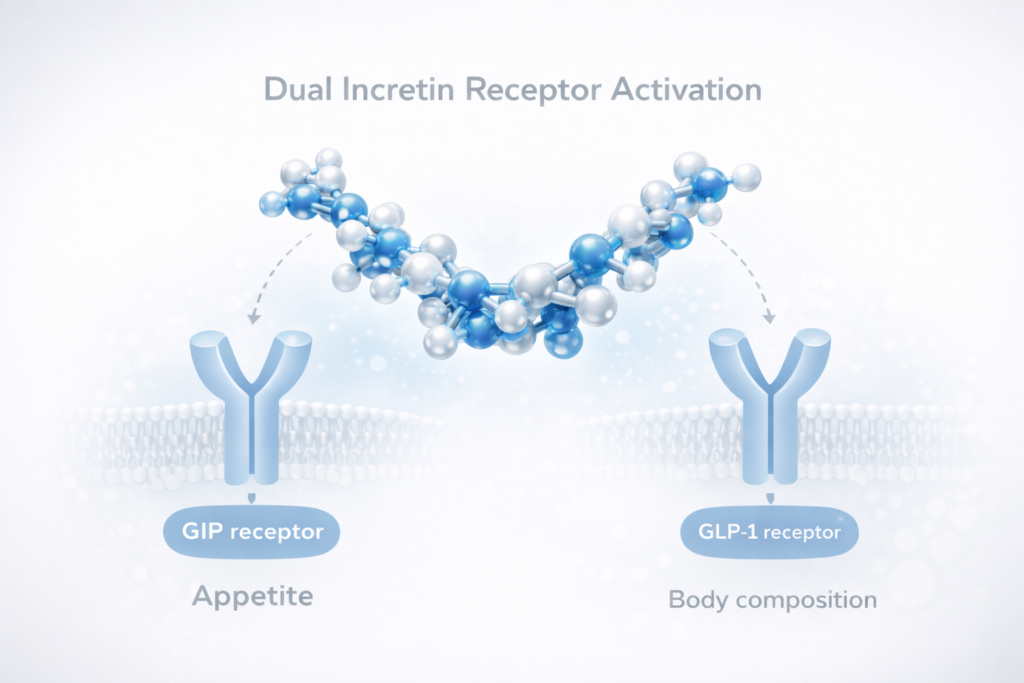
The growing prevalence of obesity and metabolic disorders has accelerated the development of incretin-based pharmacotherapies. Among these, dual incretin receptor agonist therapies represent a major scientific advancement due to their dual receptor activity targeting both glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) pathways.
Omnitida BioActive, developed by BIOTECH LABORATORIUM LTD, is an innovative medication designed for medically supervised weight management and metabolic optimisation. It aligns with modern evidence demonstrating the importance of dual incretin modulation in improving body weight, glycaemic balance, and overall metabolic health.
This article presents a professional reinterpretation of current scientific knowledge on dual incretin receptor agonist therapies, highlighting their pharmacological mechanism, clinical efficacy, and relevance in integrative metabolic and aesthetic medicine.
Sources: peer-reviewed review on dual GIP/GLP-1 receptor agonists (PMC10159347); clinical pharmacology literature on tirzepatide.
Dual Incretin Receptor Agonist
Dual incretin receptor agonist therapies involve synthetic peptide molecules engineered to activate both GIP and GLP-1 receptors, which are key hormonal regulators of glucose metabolism, appetite control, and energy homeostasis. The integration of these two incretin pathways creates a synergistic pharmacological profile that extends beyond the effects of single-pathway incretin modulation.
Activation of GLP-1 receptors primarily influences appetite suppression, delayed gastric emptying, and improved insulin secretion, whereas GIP receptor activation contributes to enhanced insulin sensitivity and direct metabolic effects on adipose tissue. The combined action leads to a broader metabolic response that targets both energy intake and energy utilisation.
This dual receptor engagement underpins the clinical effectiveness of dual incretin receptor agonist therapy in reducing body weight while simultaneously improving metabolic parameters, supporting its role as a next-generation therapeutic strategy for obesity and metabolic dysfunction.
Sources: mechanistic reviews of dual incretin receptor agonist pharmacodynamics; endocrine studies on GIP and GLP-1 signalling pathways.
Effects on Glycaemic Control and Metabolic Regulation
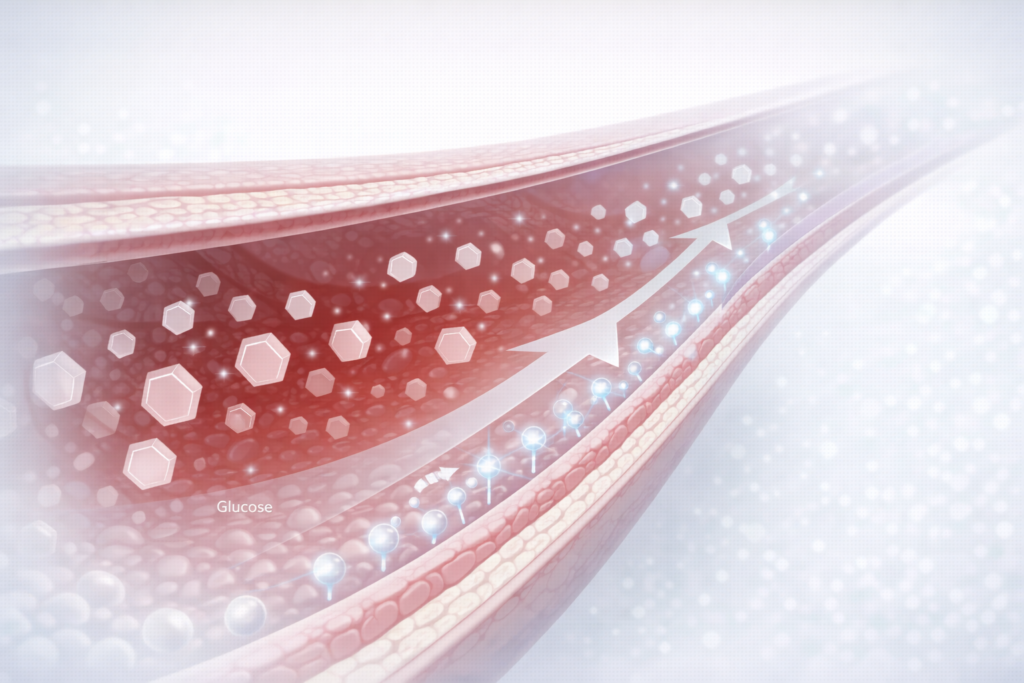
Scientific evidence demonstrates that dual incretin receptor agonist therapies significantly improve glycaemic control by enhancing glucose-dependent insulin secretion and reducing glucagon levels. These mechanisms contribute to improved insulin sensitivity and better regulation of blood glucose levels, which are closely linked to adiposity and metabolic inflammation.
The dual incretin mechanism allows this therapeutic approach to address several key components of metabolic syndrome, including impaired glucose tolerance, increased visceral fat accumulation, and dysregulated energy balance. Such comprehensive metabolic regulation is critical not only for weight reduction but also for long-term metabolic stability.
From a pharmacological perspective, this profile supports the use of dual incretin–based therapies in patients with obesity, insulin resistance, and associated metabolic disturbances.
Sources: clinical and mechanistic analyses of dual incretin receptor agonist therapies on glycaemic control; endocrinology research on incretin hormone physiology.
Clinical Impact on Body Weight and Adipose Tissue

Clinical trials and systematic analyses consistently show that dual incretin receptor agonist therapies lead to substantial reductions in body weight and waist circumference. These reductions are associated primarily with decreases in fat mass rather than lean mass, indicating favourable body composition outcomes.
In addition to overall weight loss, dual incretin–based therapy has demonstrated beneficial effects on visceral adiposity, which plays a central role in cardiometabolic risk and inflammatory processes. By reducing visceral fat stores, this therapeutic approach contributes to improvements in metabolic health that extend beyond simple numerical weight reduction.
These findings support the clinical positioning of dual incretin receptor agonist therapy as a comprehensive metabolic strategy capable of influencing both energy intake and adipose tissue biology.
Sources: randomised clinical trials evaluating dual incretin receptor agonist therapies in obesity; systematic reviews on adipose tissue and metabolic outcomes.
Relevance for Integrative Aesthetic and Metabolic Medicine
In aesthetic and dermatological practice, metabolic dysfunction is increasingly recognised as a major contributor to skin ageing, inflammatory dermatoses, and suboptimal body contouring outcomes. Excess visceral adiposity and insulin resistance can negatively influence skin elasticity, collagen quality, and inflammatory balance.
Through its dual incretin mechanism, dual incretin receptor agonist therapy supports:
- Progressive reduction of fat mass
- Improvement in insulin sensitivity
- Reduction of systemic metabolic inflammation
- Enhanced regulation of appetite and caloric intake
These systemic effects may indirectly contribute to improved aesthetic outcomes, particularly in comprehensive treatment programmes combining pharmacological therapy with dermatological and body-contouring interventions.
Sources: interdisciplinary publications on metabolic health and dermatological outcomes; reviews on obesity, inflammation, and skin physiology.
Safety Profile and Clinical Supervision
Available clinical evidence indicates that dual incretin receptor agonist therapies are generally well tolerated when used under appropriate medical supervision. The most frequently reported adverse effects are gastrointestinal in nature and are typically mild to moderate and transient, particularly during dose titration phases.
Individualised dosing and careful clinical monitoring are essential to optimise both safety and efficacy. This approach allows therapy to be tailored according to patient metabolic profile, treatment goals, and tolerance, ensuring a personalised and clinically responsible strategy.
Sources: safety analyses from clinical trials of dual incretin receptor agonist therapies; systematic reviews on incretin-based therapy tolerability.
Omnitida BioActive: Translating Scientific Evidence into Clinical Practice
Omnitida BioActive, developed by BIOTECH LABORATORIUM LTD, reflects the scientific principles demonstrated in contemporary research on dual incretin receptor agonists. By targeting both GIP and GLP-1 pathways, it provides a comprehensive pharmacological approach to weight management and metabolic optimisation.
Its therapeutic concept is aligned with modern clinical understanding that sustainable weight control requires integrated regulation of appetite, insulin sensitivity, and adipose tissue metabolism rather than isolated appetite suppression alone.
Potential clinically relevant benefits include:
- Meaningful and sustained reduction in body weight
- Improvement in glycaemic balance and insulin sensitivity
- Reduction in visceral adipose tissue
- Support for long-term metabolic stability
- Synergy with lifestyle and aesthetic treatment protocols
Conclusion
Current scientific literature highlights the importance of dual incretin receptor modulation in achieving significant improvements in body weight and metabolic health. By simultaneously targeting GIP and GLP-1 pathways, dual incretin receptor agonist therapies offer a comprehensive and physiologically grounded strategy for managing obesity and associated metabolic disorders.
Omnitida BioActive, developed by BIOTECH LABORATORIUM LTD, translates these evidence-based mechanisms into an innovative therapeutic solution suitable for integrative medical and aesthetic practice. Under professional supervision, dual incretin–based therapy represents a scientifically substantiated and forward-looking approach to effective, sustainable, and metabolically balanced weight management.
Sources: peer-reviewed review on dual incretin receptor agonists (PMC10159347); clinical trials and systematic reviews evaluating dual incretin receptor agonist therapies for efficacy, safety, and metabolic outcomes.